Soil Leaching vs. Nitrate Leaching — and Where ORP Changes the Conversation
In agronomy, leaching is often used as a catch-all term. But technically, soil (salt) leaching, nitrate leaching, and redox-driven nitrate loss are distinct processes. Conflating them leads to flawed nitrogen decisions.
Let’s separate the mechanisms, and then layer in how ORP (oxidation-reduction potential) provides insight that neither EC nor nitrate concentration alone can deliver.
Soil Leaching (Salts): Ionic Mass Movement
When we refer to soil leaching in a salinity context, we are describing:
Downward movement of total soluble ions due to water percolation.
Measured By:
- Electrical Conductivity (EC)
- Total dissolved salts (TDS)
What EC Actually Reflects:
Combined concentration of:
- Ca²⁺
- Mg²⁺
- Na⁺
- Cl⁻
- SO₄²⁻
- NO₃⁻
(and others)
EC confirms water movement and ionic redistribution.
It does not isolate nitrate behavior.
Using EC as a proxy assumes:
If salts moved, nitrate moved.
That assumption is sometimes directionally useful, but chemically incomplete.
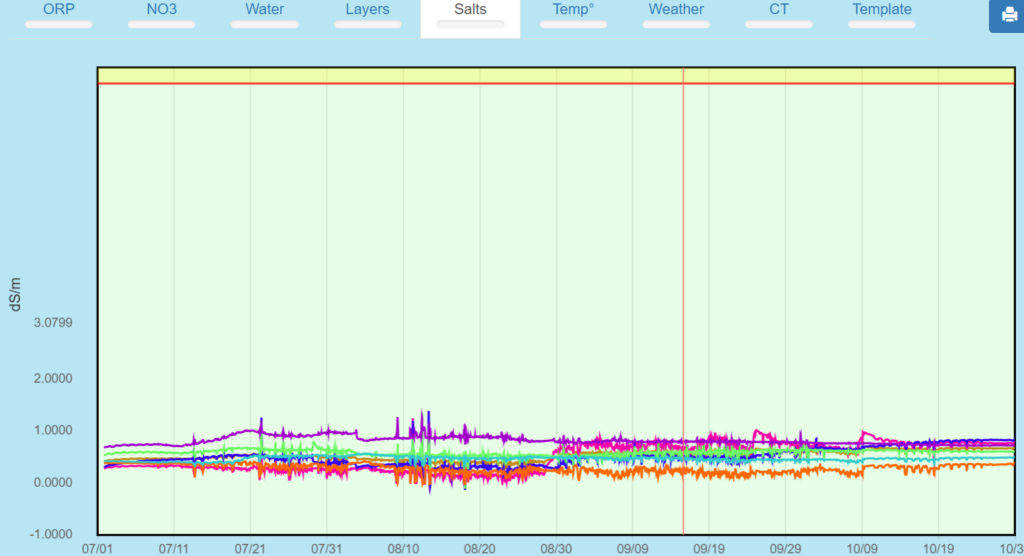
This shows no large differential in the EC by layer
Nitrate Leaching: Direct NO₃⁻-N Movement
Nitrate leaching refers specifically to:
Downward transport of NO3⁻ below the active root zone.
Nitrate is:
- Highly soluble
- Negatively charged
- Not adsorbed to soil particles
- Fully water-mobile
Measured By:
- Lab extraction
- Soil solution sampling
- Ion-Selective Electrodes (ISE) for real-time nitrate monitoring
This is direct measurement.
You know concentration, depth, and timing.
But even direct nitrate monitoring tells only part of the nitrogen story.
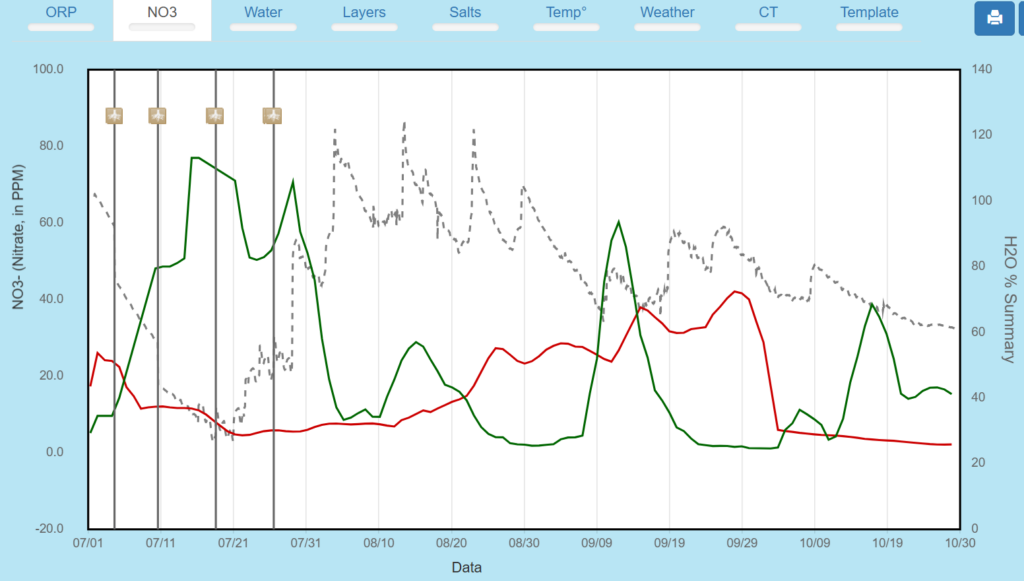
Nitrate 1 and 2 foot summary-initial N application pushed to the second foot
Where ORP Adds Critical Insight
Oxidation-Reduction Potential (ORP) measures the soil’s electron activity — essentially its oxygen status.
ORP does not measure nitrate.
But it determines what nitrate can become.
High ORP (oxidized soil):
- Oxygen present
- Nitrification favored
- Nitrate stable
- Loss risk primarily = leaching
Low ORP (reduced / anaerobic soil):
- Oxygen depleted
- Denitrification favored
- Nitrate converts to N₂O and N₂ gases
- Loss risk = gaseous, not leaching
This is the critical distinction:
Not all nitrogen loss is leaching.
Some nitrogen disappears biologically as soil organic matter before it ever leaches.
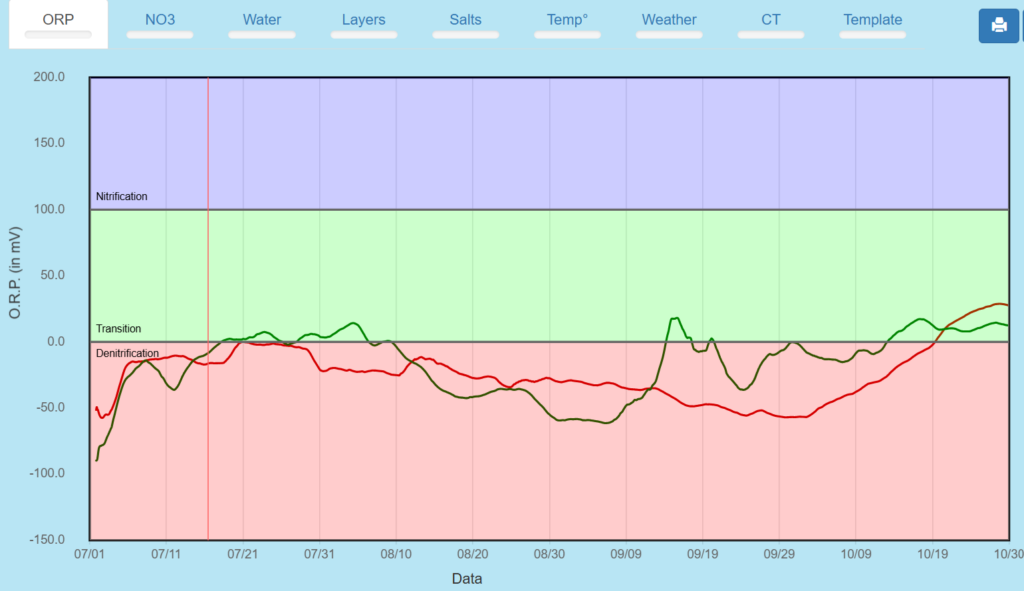
ORP 1 and 2 foot summary – mostly in denitrification
Leaching vs. Denitrification: Mechanistic Divergence
Scenario A: High EC + Nitrate Decline + High ORP
→ Likely downward movement (true leaching)
Scenario B: Nitrate Decline + Low ORP + No Deep Nitrate Spike
→ Likely denitrification (gaseous loss)
Scenario C: EC Stable + Nitrate Stable + Low ORP
→ Nitrate temporarily immobilized or at risk of reduction
Without ORP, nitrate decline is ambiguous.
With ORP, mechanism becomes interpretable.
Proxy vs. Direct + Process Insight
| Measurement Type | What It Tells You | What It Cannot Tell You |
| EC (Salts) | Water & total ion movement | Which ion moved |
| Nitrate ISE | Exact NO3⁻ concentration | Why nitrate changed |
| ORP | Soil oxygen status & redox pathway | Nitrate concentration |
Combined Interpretation:
- EC → Did water move?
- Nitrate → Did nitrogen move?
- ORP → Was nitrogen chemically stable?
Together, they transform nutrient management from reactive to diagnostic.
Why This Matters Agronomically
If nitrate concentration drops, three mechanisms are possible:
- Leaching below roots
- Plant uptake
- Denitrification
EC alone cannot distinguish them.
Nitrate alone cannot distinguish biological reduction from physical movement.
ORP provides the biochemical context.
In saturated, low-ORP conditions, nitrate loss may occur without measurable leaching.
Conversely, in high-ORP conditions with strong percolation, nitrate loss is dominantly hydrologic (leaching).
Terminology Alignment
To be technically precise:
- Soil leaching (salts) = bulk ionic movement (EC-based inference)
- Nitrate leaching = measured NO₃⁻ transport below root zone
- Redox-driven nitrate loss = denitrification under low ORP
Leaching is hydraulic.
Denitrification is biochemical.
ORP distinguishes the two.
Closing Perspective
Water drives movement.
Redox governs transformation.
Measurement determines certainty.
Using salts as a proxy is inferential.
Using nitrate sensors is confirmatory.
Adding ORP makes it diagnostic. In advanced nitrogen management, that distinction is not academic — it is economic and environmental.

